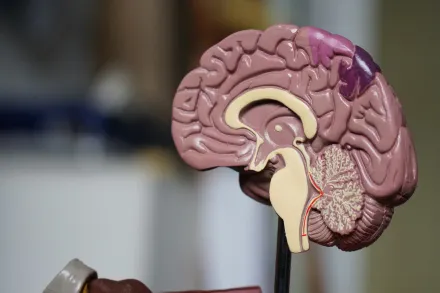
Epilepsy is a complex neurological condition defined by a tendency to experience recurrent seizures that arise from abrupt bursts of electrical activity within the brain. The phenomenon is not a single disease with a single cause but rather a spectrum of conditions in which the brain becomes prone to excessive synchronous discharges. The path from a normal brain to a brain that repeatedly generates seizures can be subtle and multifaceted, often involving a combination of genetic predispositions, structural alterations, metabolic disturbances, immune system interactions, and environmental influences. The study of epilepsy seeks to unravel how these factors interact over a person’s lifespan, sometimes starting before birth, sometimes developing after an injury or illness, and occasionally emerging without an obvious trigger. The diversity of etiologies means that two individuals with the same diagnosis may have remarkably different underlying mechanisms, therapies, and prognostic considerations, underscoring the importance of personalized assessment and care.
Understanding the causes of epilepsy begins with recognizing that the brain is an intricate network of neurons communicating through precise electrical impulses. When this communication becomes irregular or unstable, seizures can occur. In some cases, seizures reflect a localized region of scar tissue, malformation, or inflammation that disrupts the normal balance of excitation and inhibition. In others, systemic factors such as metabolic disorders, autoimmune processes, or infectious diseases may lower the threshold for seizures across widespread brain regions. The natural history of epilepsy often includes periods of relative stability punctuated by episodes triggered by stressors like sleep deprivation, illness, alcohol withdrawal, or fever in children. Yet many events happen without any identifiable trigger, which is why clinicians emphasize a careful history, neurological examination, and targeted testing to determine the most probable causes and appropriate treatment pathways. The following sections explore the major categories of epilepsy causes in a comprehensive, cohesive manner, with attention to how these categories manifest in clinical practice and what they imply for management and outcomes.
Genetic and familial factors
Genetic influences play a substantial role in many forms of epilepsy, ranging from highly specific single gene mutations to broader inherited susceptibility patterns that elevate the risk of seizures across generations. In certain syndromes, particular gene variants reliably produce epileptic activity by altering how brain cells generate, transmit, or regulate electrical signals. These genetic changes can affect ion channels, neurotransmitter receptors, or the machinery responsible for neuronal development, synaptic formation, and network connectivity. The consequences may appear early in life, during infancy or childhood, or may remain latent until later when environmental or developmental changes reveal the underlying vulnerability. It is important to recognize that possessing a genetic predisposition does not guarantee that seizures will occur; rather, it increases the likelihood that the brain's delicate balance between excitation and inhibition can be disrupted under certain circumstances.
In some cases, a specific genetic diagnosis is established by modern sequencing techniques, revealing mutations in genes that code for ion channels or synaptic proteins. These discoveries not only explain why seizures arise but also guide treatment decisions in certain syndromes where particular medications or therapies have demonstrated superior efficacy. Beyond single gene mutations, a broader heritability pattern exists in which families show clustering of epilepsy across generations without a single dominant abnormal gene being identified. This complexity reflects the polygenic nature of brain excitability, where multiple modest genetic factors interact with each other and with environmental influences to shape seizure risk. Clinicians consider family history, age at onset, seizure types, and associated neurological features when contemplating a genetic contribution, and genetic counseling can provide valuable information for families planning future children or seeking reassurance about recurrence risk.
Structural brain abnormalities
Structural abnormalities refer to physical alterations in the brain's anatomy that disrupt normal circuitry and create an environment conducive to epileptogenic activity. These changes can be congenital, meaning present from birth due to developmental disturbances, or acquired later in life as a result of injury, infection, or disease processes. When abnormal brain tissue forms a focal region where neurons fire in a synchronized and excessive manner, seizures may originate in that spot and spread to surrounding networks. Examples include cortical malformations arising from disrupted neuronal migration during development, scar tissue from prior injuries, scars after infections, and brain tumors that alter local or network dynamics. Even small, subtle malformations can be enough to generate recurrent seizures, underscoring the brain's sensitivity to architectural organization and synaptic balance. In some cases, imaging studies such as magnetic resonance imaging reveal these structural alterations, while in others, a comprehensive evaluation including EEG monitoring helps pinpoint regions of the brain that are prone to seizure activity.
Injuries and surgical lesions can also lead to new-onset epilepsy years after the initial event. When an area of the brain sustains damage, scar tissue may form and create a persistent focus that remains electrically excitable. This process can happen after a traumatic brain injury, a stroke, an infection, or an inflammatory event that leaves residual changes in neuronal connections. Epilepsy that arises from such structural problems often features seizures that begin in a defined region and may be resistant to standard medical therapy, prompting consideration of specialized treatments such as targeted therapies or surgical interventions aimed at removing or disconnecting the epileptogenic tissue while preserving vital brain functions. The interplay between focal pathology and network-wide brain activity is a central topic in the understanding and management of structural epilepsy, guiding decisions about diagnostic testing, prognosis, and treatment strategy.
Metabolic and biochemical influences
Metabolic disturbances can alter the balance of ions, energy supply, and neurotransmitter systems that govern neuronal excitability. When metabolic processes falter or push beyond the brain’s compensatory capacities, seizures may emerge as a consequence of neuronal stress or impaired restoration of normal electrical activity after episodes of abnormal firing. Conditions such as severe electrolyte imbalances, low blood sugar, or disturbances in calcium, magnesium, and other minerals can transiently raise seizure susceptibility. Inborn errors of metabolism, though rarer, can lead to the accumulation of abnormal substances or deficiencies in critical cofactors that disrupt brain metabolism and promote epileptogenic networks. The developing brain may be particularly sensitive to metabolic insults, which can shape seizure patterns that persist into adulthood if not addressed promptly and comprehensively.
A subset of metabolic epilepsy arises from mitochondrial disorders, where the energy-producing compartments within cells fail to function properly. Neurons require a constant and robust energy supply, and when mitochondrial efficiency is compromised, the resulting energy crisis can provoke abnormal electrical activity across neural circuits. Treating these conditions involves a combination of disease-specific strategies and seizure management that accounts for the broader systemic implications. In addition to energy metabolism, dysregulation of neurotransmitter systems, including GABAergic and glutamatergic signaling, can tilt the balance toward hyperexcitability. Metabolic contributors to epilepsy remind clinicians that the brain does not operate in isolation from body-wide physiology, and addressing the underlying metabolic condition can be a crucial part of reducing seizure frequency and improving quality of life.
Infections and inflammatory processes
Infections of the brain and surrounding membranes, as well as inflammatory conditions that influence neural tissue, are important causes of epilepsy, particularly in children and young adults but also in older adults under certain circumstances. Infections such as meningitis, encephalitis, brain abscesses, and parasitic or other infectious processes can damage delicate neural networks, alter the neuronal milieu, and ignite persistent hyperexcitability. Even after the acute infection resolves, scar formation or ongoing inflammatory activity may sustain seizure risk by modifying the local circuitry and its threshold for synchronous discharge. Autoimmune epilepsies, in which the immune system mistakenly targets brain components, represent another dimension of inflammatory epilepsy. In these cases antibodies or immune cells attack neuronal receptors or synaptic proteins, leading to seizures that may be part of broader autoimmune syndromes or isolated brain inflammation. The treatment implications are distinct, often requiring immunomodulatory therapies alongside conventional antiseizure medications to address the underlying immune-driven process.
The brain’s response to infection or inflammation can vary widely among individuals, depending on factors such as age, genetic predisposition, the site of involvement within the brain, and the timeliness of medical intervention. Clinicians assess for signs of inflammatory activity through clinical evaluation, laboratory studies, imaging, and sometimes autoimmune testing. The management of infection-associated or inflammatory epilepsy emphasizes rapid treatment of the underlying cause, control of seizures with appropriate medications, and strategies to minimize long-term neural disruption. This area of epilepsy highlights the dynamic relationship between the immune system, the brain’s structural integrity, and electrical stability, illustrating why a multidisciplinary approach is often essential for optimal outcomes.
Head trauma and injuries
Traumatic brain injury is a well-recognized precipitant of epilepsy, and its effects can appear soon after the injury or emerge years later as delayed changes in brain tissue evolve. The initial insult can injure neurons, disrupt protective barriers, and set in motion cascades of inflammation, edema, and scarring that collectively alter how brain networks communicate. Even injuries that might seem minor at first can create lasting alterations in neural circuitry, particularly when they affect regions prone to generating seizures, such as the temporal lobes. Recurrent post-traumatic seizures may be linked to the location and severity of the injury, the presence of hemorrhage or contusion, and the brain’s capacity to recover or reorganize after damage. In some individuals, brain trauma also changes the delicate balance between excitatory and inhibitory neurotransmission, increasing the likelihood that normal neuronal activity can become pathologically synchronized and propagate into a seizure event.
Management implications for trauma-related epilepsy include careful assessment of injury history, imaging to identify enduring lesions, and a tailored antiseizure medication plan that accounts for potential comorbidities such as cognitive impairment, mood changes, or motor dysfunction. In certain cases, neurosurgical approaches or neuromodulation therapies may be considered when seizures originate from a specific brain region that can be safely targeted. The long-term outlook depends on factors like seizure control with medications, the extent of tissue scar, and how well the brain adapts after injury, all of which guide ongoing monitoring and adjustments in therapy over time.
Stroke and vascular factors
Strokes, whether ischemic or hemorrhagic, disrupt the blood supply to brain tissue and can leave behind areas of irreversibly damaged tissue that become foci for epileptic activity. The occurrence of seizures after a stroke may be immediate or delayed, with the risk increasing in the weeks to months following the event. The brain’s response to vascular injury involves inflammation, cell death, and remodeling of neural networks, and these processes can create new pathways for abnormal electrical discharges to arise and propagate. In older adults, vascular risk factors such as high blood pressure, atherosclerosis, and metabolic disorders can contribute to a higher incidence of post-stroke epilepsy, though the condition can also appear in younger individuals when vascular injury occurs due to unusual causes or genetic predispositions. The management of vascular epilepsy emphasizes rapid stabilization, prevention of further vascular events, and optimization of seizure control through medication choices that consider interactions with anticoagulants, blood pressure targets, and overall cardiovascular health.
Beyond focal strokes, other vascular conditions that disrupt perfusion and tissue integrity may also lead to seizures. Microbleeds, large vessel disease, and certain congenital vascular malformations can create focal irritative zones or promote widespread network instability. The interplay between vascular health and neural excitability is a reminder that systemic conditions, including diabetes and cholesterol management, can indirectly influence seizure risk by shaping the brain’s resilience to insults and its capacity to recover proper electrical balance after a vascular event. Clinicians integrate imaging findings, patient history, and functional assessments to map the likely epileptogenic regions and to determine the best approach for reducing seizure burden while preserving cognitive and motor function.
Prenatal and perinatal factors
Events that occur before birth and around the time of birth can lay the groundwork for epilepsy that becomes apparent early in life or even later in childhood. Prenatal factors such as maternal infections, exposure to toxins, insufficient oxygen supply during birth, or complications that affect fetal development can influence brain formation and the establishment of normal cortical architecture. When subtle or overt disturbances in neuronal migration or synaptic organization occur during critical windows of brain development, the resulting networks may be predisposed to epileptogenic activity as the child grows. Perinatal complications, including premature birth, birth asphyxia, or metabolic disturbances in the newborn period, can set the stage for seizures by altering neuronal excitability and the maturation of inhibitory circuits that normally help restrain excessive firing. The consequences of these early events are often influenced by the child’s broader medical history, the presence of other congenital anomalies, and the quality of neonatal care that supports development and healing in the first days and weeks of life.
Early-life seizures may themselves contribute to adjusting neural networks in ways that impact future epilepsy risk, a concept that underscores the importance of rapid and comprehensive evaluation when seizures occur in infancy. In some cases, genetic factors interact with prenatal or perinatal insults to create a compounded risk, illustrating the intricate interplay between inherited predispositions and early environmental exposures. Understanding these factors helps clinicians counsel families about prognosis, plan early interventions, and coordinate multidisciplinary care that supports development while addressing potential seizure disorders as the child grows toward adolescence and adulthood.
Developmental disorders and cortical malformations
Developmental disorders involving cortical development—such as malformations arising from disrupted neuronal migration, abnormal cortical layering, or irregular organization of the brain’s gray matter—are important contributors to epilepsy in many individuals. These malformations can be congenital, present from birth, or develop in early childhood as the brain matures. The exact appearance of these abnormalities on imaging studies depends on the specific pattern of disruption, but the common thread is that abnormal cortical structure can create aberrant electrical circuits that serve as sources of epileptic activity. Seizures associated with cortical malformations often have particular semiologies that reflect the region of the brain affected, and they may persist despite standard antiseizure medications, requiring specialized diagnostic and therapeutic planning. The relationship between structure and function in the brain makes these conditions a central focus of discussions about why epilepsy arises and how best to tailor interventions to individual patients.
In addition to cortical malformations, other developmental issues such as tuberous sclerosis, focal cortical dysplasia, and lissencephaly illustrate how early neurodevelopmental processes shape lifelong seizure risk. Each condition carries its own profile of associated features, including cognitive and behavioral implications, that influence overall care. Clinicians use high-resolution imaging, developmental assessments, and genetic testing when appropriate to delineate the specific cause and to discuss prognosis with families. Although structural causes can be challenging to manage, advances in surgical techniques, neuromodulation, and adaptive therapies offer meaningful options for select patients, aiming to reduce seizures while preserving or improving neurological function and quality of life.
Autoimmune and immune-mediated etiologies
Autoimmune mechanisms are increasingly recognized as a source of epilepsy, particularly when seizures occur in patterns that respond to immunotherapy or coincide with other signs of immune system activation. In autoimmune epilepsies, antibodies or immune cells target neuronal surface receptors, synaptic proteins, or other components of the nervous system, leading to disruptions in synaptic transmission and network instability. The presentation of autoimmune epilepsy can include rapid onset of seizures, associated cognitive changes, psychiatric symptoms, movement disorders, or symptoms indicating brain inflammation. Diagnostic evaluation in these cases often involves testing for specific autoantibodies, inflammatory markers, and careful clinical correlation to identify an immune-driven process that may require immunomodulatory treatment in addition to conventional antiseizure therapy. The recognition of autoimmune etiologies has expanded treatment options and improved outcomes for patients who once faced limited prospects for seizure control.
Autoimmune epilepsy underscores the brain’s vulnerability to immune dysregulation and the way systemic immune processes can translate into localized neural excitability. Because immune-mediated seizures may respond dramatically to timely intervention, early suspicion and comprehensive evaluation are crucial. The approach to management is multidisciplinary, inviting collaboration among neurologists, immunologists, endocrinologists, and psychiatrists to address the complex medical and psychological needs that often accompany autoimmune conditions. This collaboration helps tailor therapies to reduce inflammation, protect neural tissue, and restore a healthier balance of neural network activity, with attention to minimizing side effects and maximizing functional recovery.
Idiopathic epilepsy and unknown causes
Not all epilepsy can be traced to a single identifiable cause. A substantial portion of cases are categorized as idiopathic, meaning that the available information does not reveal a clear structural, metabolic, genetic, infectious, or autoimmune driver. In these instances, seizures arise in individuals with normal MRI scans and no obvious etiologies despite thorough evaluation. Idiopathic epilepsy often has a predominant age of onset and characteristic seizure types that guide management and prognosis. Even when the precise mechanism remains elusive, the clinical picture informs treatment choices, including the selection of antiseizure medications and nonpharmacological strategies such as lifestyle adjustments that can substantially reduce seizure frequency and improve daily functioning. Ongoing research continues to illuminate subtle mechanisms that might account for idiopathic cases, including unidentified genetic factors, microstructural brain differences, and dynamic brain network properties that only become evident with advanced neuroimaging and long-term monitoring.
The category of unknown or idiopathic epilepsy highlights the limits of current knowledge while emphasizing a practical approach to care. Clinicians focus on achieving effective seizure control, minimizing adverse effects, and supporting cognitive, emotional, and social development. Family engagement, patient education, and regular follow-up help track changes over time and adapt treatment plans as new information or therapies become available. This realm of epilepsy care acknowledges that even without a definitive cause, meaningful progress can be achieved through careful management grounded in individualized assessment and evidence-based practice.
Environmental and lifestyle contributors
Environmental and lifestyle factors can modulate seizure susceptibility in people with epilepsy and may influence the course of the condition even when a primary cause is already identified. Sleep deprivation, irregular sleep patterns, stress, alcohol use, certain illicit substances, and exposure to specific chemicals or toxins can lower the seizure threshold or precipitate breakthrough seizures in susceptible individuals. While these factors do not create epilepsy in most cases, they can shape the frequency and severity of seizures and therefore become important targets for counseling and behavioral modification as part of a comprehensive treatment plan. Education about sleep hygiene, stress reduction, and avoidance of known triggers complements pharmacologic therapy and supports overall well-being. Families and patients often find it helpful to keep symptom diaries that track seizure occurrence in relation to sleep, illness, medication adherence, and lifestyle changes, providing practical insights that inform personalized strategies for reducing seizure burden.
Healthcare providers also emphasize the importance of safety measures and proactive management in daily life for people living with epilepsy. Understanding triggers helps patients plan activities, engage in exercise, and participate in social and occupational endeavors with confidence. A holistic approach that includes sleep optimization, nutrition, physical activity, mood management, and regular medical checkups contributes to better seizure control and a higher quality of life. Although the path to understanding epilepsy causes is complex and multifaceted, this approach empowers individuals to participate actively in decisions about treatment, monitoring, and support systems, while researchers continue to uncover the nuanced mechanisms that underlie this diverse neurological spectrum.