Lung cancer often grows silently and presents symptoms only after it has advanced, which makes timely detection crucial. Readers who are attentive to changes in breathing, cough patterns, or chest sensations can gain an opportunity to seek evaluation before disease progresses. The goal of this guide is to present a clear, patient centered overview of early signals and practical steps to pursue care in a responsible, informed way. By understanding the signs, risk factors, and available screening options, individuals can participate more actively in their health without becoming overwhelmed by fear.
Across populations, awareness of early signals can empower people to act promptly while avoiding excessive anxiety. This examination also highlights why clinicians stress a careful, personalized approach, because symptoms that resemble other respiratory conditions may mask early cancer and require careful assessment rather than immediate alarm. The interplay between symptoms, risk history, and diagnostic tests shapes the path toward confirmation or reassurance in a way that respects patient experience and medical caution.
In this discussion, the focus remains on clear, evidence informed information about what to notice, when to seek care, and how modern screening tools fit into an overall strategy for detecting lung cancer as early as possible. It is important to acknowledge that not all signs mean cancer, and many benign conditions can produce similar sensations or coughs. The emphasis is on recognizing patterns that persist, worsen, or appear in people with higher risk, and on engaging with healthcare providers to distinguish temporary irritants from something that warrants further evaluation.
Understanding the landscape of lung cancer
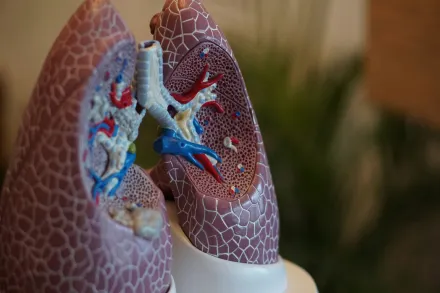
Lung cancer is a disease of the respiratory system that can originate in different cell types, with some forms growing more rapidly than others. The pace of growth, the location of the tumor, and the spread of cancer cells influence the timing of symptoms and the likelihood of successful early detection. While modern treatment options have advanced significantly, the best outcomes are often achieved when cancer is identified before it has spread beyond the lungs. This is why screening programs and public awareness campaigns emphasize vigilance for persistent respiratory changes, even when they seem mild at first glance.
Factors that contribute to the development of lung cancer include environmental exposure, lifestyle choices, and genetic predisposition. Smoking remains the primary modifiable risk factor, but long term exposure to secondhand smoke, radon gas, occupational carcinogens such as asbestos, and air pollution also contribute to risk. Additionally, certain genetic markers and family histories can influence an individual’s likelihood of developing cancer at a younger age or in the absence of traditional risk factors. Understanding these nuances helps clinicians tailor screening recommendations and helps patients assess their personal risk more accurately.
The clinical landscape also includes a spectrum of disease stages, with early stage cancers generally confined to the lungs and later stages showing spread to lymph nodes or distant organs. The early signs are often subtle and easily mistaken for viral infections, bronchitis, or chronic conditions like asthma. Recognizing how these signals might relate to underlying pathophysiology can improve the likelihood that a person seeks timely evaluation rather than attributing symptoms to a temporary condition. Through education and shared decision making, patients can participate more confidently in conversations about screening and diagnostic testing with their doctors.
Early signs and symptoms to watch for
A persistent cough that lasts for weeks, especially one that does not respond to usual remedies, is among the most common early clues. A cough may be new or have changed in character, becoming more intense or producing mucus that has a different color or consistency than before. This change should prompt a conversation with a healthcare professional, particularly if it is accompanied by other signs described below. Coughing up blood, even in small amounts, is not something to ignore and requires prompt medical evaluation to determine the cause and to rule out cancer or other serious conditions.
Shortness of breath or a sense of chest tightness that appears without an obvious trigger may signal a problem with the lungs or surrounding structures. Breathlessness can be subtle at first, occurring with exertion or after activities that previously did not provoke symptoms. The presence of wheezing, hoarseness, or a feeling of fullness in the chest can also accompany early disease or reflect irritation of the airways and nerves near the tumor. Any new, persistent breathing difficulty warrants careful assessment rather than assuming it is related to a temporary infection or deconditioning.
Chest pain that feels dull, aching, or persistent can be a sign that a tumor is pressing on nerves, ribs, or other tissues in the chest. Pain associated with coughing or deep breathing may be more pronounced in the presence of a tumor, and it should be evaluated promptly to determine whether the origin is benign or malignant. Recurrent respiratory infections, such as bronchitis or pneumonia that repeatedly return despite treatment, can signal an underlying process that merits investigation. When infections recur in the same part of the lung, doctors may look more closely for a structural cause, including the possibility of a tumor.
Unexplained and unintentional weight loss, loss of appetite, or persistent fatigue are signals that the body may be dealing with a systemic process. These nonspecific signs can accompany many conditions, but when they occur alongside respiratory symptoms or risk factors, they warrant a medical workup. Some individuals experience a change in voice, such as hoarseness, which can occur when cancer affects the nerves controlling the vocal cords or the airway structures. Being attentive to these changes can help people initiate timely discussions with their clinicians.
Beyond the classic signs, some people notice changes in their nails or skin, a sense of fullness in the chest, or recurrent coughing with production of mucus that contains blood or appears darker than usual. While these signs do not establish a cancer diagnosis, they contribute to the overall clinical picture considered during evaluation. It is essential to tell the clinician about all symptoms, even those that may seem minor, because collectively they shape the next steps in testing and referral decisions.
Many early cancers do not cause symptoms at all and are detected incidentally during imaging performed for other reasons. In such scenarios, a clinician will review whether preliminary signs, family history, or risk exposure justify additional testing. For individuals without symptoms but with elevated risk, screening programs can identify cancer earlier than symptom driven detection, offering meaningful improvements in prognosis when adhered to appropriately.
When symptoms warrant medical evaluation
If any warning signs persist for more than a few weeks, a medical evaluation becomes prudent. A thorough history and physical examination, accompanied by discussion of risk factors like smoking, occupational exposures, and family history, helps shape the decision to pursue imaging studies. Early referral to a clinician with expertise in lung diseases can facilitate a careful and timely assessment, including consideration of chest imaging, laboratory tests, and potential referrals to specialists for further evaluation.
In cases where symptoms appear suddenly and are severe, such as chest pain with shortness of breath or coughing up a significant amount of blood, immediate medical attention is warranted. While these scenarios may be frightening, they necessitate urgent evaluation to exclude life threatening conditions and to determine whether cancer could be a contributing factor. The goal of prompt assessment in these situations is to confirm or rule out serious etiologies and to initiate appropriate treatment without delay if cancer is suspected.
Individuals who have a higher level of risk due to age, smoking history, or environmental exposures should consider proactive discussions with their healthcare provider about screening and diagnostic options even when symptoms are not obviously present. In many cases, clinicians will propose surveillance strategies or noninvasive tests to monitor changes over time, enabling earlier detection if a developing cancer begins to make itself known through shifts in imaging or symptoms.
Screening and detection methods
Screening for lung cancer primarily relies on imaging, with low dose computed tomography, known as LDCT, playing a central role for certain high risk groups. LDCT uses a lower amount of radiation than a standard CT, making it suitable for routine screening in people who are at increased risk because of age and smoking history. Screening programs aim to identify tumors when they are small and more manageable, often before noticeable symptoms emerge, which can substantially improve treatment outcomes and survival chances. It is important to understand that screening is not a diagnostic test; any suspicious finding requires further confirmation with diagnostic workups.
Eligibility criteria for screening typically focus on adults in mid to late adulthood who have a substantial smoking history and who have a reasonable life expectancy and ability to undergo treatment if needed. Clinicians may also consider other risk factors such as exposure to radon or occupational carcinogens and family history when discussing screening. The decision to undergo LDCT should involve a shared decision making process, in which patients are informed about potential benefits, risks such as false positives, and possible follow up procedures, and then decide together with their clinician.
In addition to LDCT, some individuals may undergo chest radiography or magnetic resonance imaging in specific circumstances, but these modalities are less sensitive for detecting early lung cancers compared with LDCT. The role of ultrasound is limited in this context and is not routinely used for initial lung cancer screening. When imaging detects a suspicious lesion, the next steps often involve a structured pathway that includes repeat imaging to monitor growth, or a referral for biopsy to obtain a tissue diagnosis and to determine exact histology, which guides treatment planning.
What happens during a CT scan
A CT scan is a fast, noninvasive procedure that provides detailed cross sectional images of the chest. For screening purposes, the LDCT protocol uses minimal contrast and a lower radiation dose compared with diagnostic CT studies. Patients are typically asked to lie still while the scanner makes multiple passes around the chest. The procedure is painless, and most people resume normal activities immediately afterward. Some centers may require a brief follow up visit to share results and discuss next steps, which will depend on what the images show and the clinical context.
The interpretation of CT images focuses on identifying nodules, masses, and other abnormalities in the lung tissue. Radiologists assess the size, shape, location, density, and growth pattern of any detected lesions. A key concept in screening is the assessment of growth over time, because small nodules may remain stable for months or years, while others may change in ways that raise concern for cancer. The radiologist will categorize findings according to established guidelines and recommend appropriate management, which can range from watchful waiting with periodic scans to immediate investigation with biopsy or surgical planning.
Radiation exposure is a consideration in any imaging study, but the risk from LDCT is typically outweighed by the potential benefits of early cancer detection for individuals at high risk. Healthcare teams emphasize careful communication about benefits and potential harms, including false positives that may lead to additional procedures, anxiety, and unnecessary tests. To minimize unnecessary interventions, clinicians use risk stratification, standardized reporting, and multidisciplinary discussions to guide the next steps after a screening chest image is obtained.
Interpreting imaging findings
When a lesion is detected, the radiologist describes its characteristics and compares it to prior scans if available. Small nodules that are less than a certain size may be classified as low risk and monitored with short term follow up imaging, while larger nodules or those with suspicious density or irregular borders may prompt more immediate action. The growth rate is particularly informative; nodules that double in size within a certain interval are more likely to be malignant, but even slow growing nodules require careful evaluation in the overall clinical context.
A chest imaging result often triggers a sequence of decisions involving primary care physicians, pulmonologists, or thoracic surgeons. The team evaluates the likelihood of cancer from imaging alone and may discuss the need for tissue sampling. Noninvasive strategies, such as certain imaging features and advanced algorithms, can sometimes reduce unnecessary biopsies, but tissue confirmation remains the gold standard for a definitive diagnosis. The goal is to balance timely detection with the least invasive approach that yields reliable information about the presence of cancer and its stage.
Ultimately, imaging is a critical step in the detection process, but it does not provide a complete answer. A convincing radiologic suspicion must be followed by pathology to confirm cancer and to guide treatment planning. Clinicians explain results in the context of the patient’s symptoms, risk factors, and overall health, ensuring that patients understand why further steps are recommended and what each step aims to achieve. This collaborative approach helps patients feel informed and supported as they navigate potential next steps with confidence.
Biopsy and pathology: confirming the diagnosis
When imaging raises concern for cancer, physicians often pursue a tissue diagnosis to confirm malignant cells and to determine tumor type. Biopsy procedures vary depending on lesion location and accessibility, and may involve guiding needles through the chest wall, bronchoscopy to access the airways, or surgical sampling in some cases. The collected tissue is sent to a pathology lab where specialists examine cells under a microscope and perform ancillary tests to determine histology, aggressiveness, and potential molecular targets for therapy.
Getting a definitive diagnosis is a turning point that shapes the entire treatment plan. Pathology reports provide essential information such as the distinction between small cell and non small cell lung cancer, the presence of specific mutations, and markers that predict response to targeted therapies. In some cases, additional tests like immunohistochemistry or genomic profiling are necessary to identify mutations or rearrangements that can be targeted with modern medicines, enabling a more personalized approach to care. Understanding these details can feel overwhelming, but clinicians are trained to interpret results and translate them into clear next steps for the patient.
Because some cancers grow within the lung without causing obvious symptoms, timely biopsy after suspicious imaging can significantly influence prognosis. A rapid, coordinated pathway between imaging specialists, interventional radiologists, pulmonologists, and pathologists helps minimize delays and ensures that patients receive accurate information about their disease status sooner rather than later. Even when a lesion is benign, the diagnostic process often provides reassurance and helps tailor follow up to each individual situation, reducing unnecessary worry and focusing attention on genuine concerns.
Biomarkers and non imaging approaches
Beyond tissue diagnosis, researchers continually explore biomarkers that might aid early detection or monitoring of lung cancer. Blood-based tests, urine analyses, and circulating tumor DNA studies represent areas of active investigation, aiming to detect cancer signals in a minimally invasive way. While these approaches hold promise, they remain supplementary to imaging and pathology in routine clinical practice for most patients. Clinicians describe these tools as potential adjuncts that could someday improve screening accuracy or monitor treatment response more conveniently.
Non imaging methods can also help in understanding risk and guiding decisions. For instance, spirometry and pulmonary function tests may reveal the impact of a growing tumor on lung performance, and they can support clinicians in interpreting imaging findings within the broader framework of a patient’s functional status. While these measures do not replace imaging, they contribute to a comprehensive assessment and help tailor interventions to preserve quality of life and maintain physical capacity during treatment and recovery.
Ultimately, the combination of imaging, pathology, and emerging biomarkers shapes an integrated approach to lung cancer detection. Patients benefit from a clear explanation of what each piece of information means, how the pieces fit together to establish a diagnosis, and what options exist for treatment planning. This collaborative, transparent process supports informed choices and aligns clinical aims with patient values and preferences, which is especially important in a disease that often unfolds over months or years.
Risk factors beyond smoking
Although smoking is the most well known risk factor for lung cancer, inhaled toxins with long term exposure can raise risk even in non smokers. Radon, an invisible gas that can accumulate in homes, is a leading environmental cause of lung cancer in nonsmokers in many regions. Occupational exposures, including asbestos, diesel exhaust, heavy metals, and certain chemical solvents, contribute to risk and underscore the importance of protective measures in the workplace and appropriate air quality controls in buildings. Indoor and outdoor air pollution are additional factors that can influence risk over time, particularly in densely populated or industrial areas.
A family history of lung cancer can increase risk even for individuals who have never smoked, suggesting a genetic component that interacts with environmental exposures. Certain inherited genetic variations may make the lung tissue more susceptible to malignant transformation when challenged by harmful substances. Understanding this interplay helps clinicians refine risk assessment and can influence screening discussions for people who might not meet typical smoking based criteria yet carry other risk markers.
Age remains a strong correlate with incidence, as risk accumulates with longer exposure to carcinogens and with natural aging processes that can alter cellular repair mechanisms. While screening guidelines address age ranges to optimize benefit, clinicians consider personal health status, comorbidities, and life expectancy when making recommendations about screening for older adults or for those who have recently stopped smoking but carry other risk factors. Education about diverse risk profiles supports inclusive conversations about lung cancer detection across different communities and life circumstances.
Prevention and reducing risk
The most effective strategy to reduce the burden of lung cancer is to avoid or stop smoking, a change that can dramatically lower risk over time. Quitting smoking at any age yields meaningful health benefits, and many programs provide resources, counseling, and pharmacologic aids that increase the chances of successful cessation. In addition to avoiding tobacco, addressing radon exposure through home testing and remediation can further reduce risk. Radon is colorless and odorless, so testing is essential for homes where levels may be elevated, especially in regions with known geological predispositions.
Mitigating occupational exposures through protective equipment, ventilation improvements, and adherence to safety guidelines reduces the risk for workers in high hazard industries. Regular maintenance of air quality in indoor environments, including the use of high efficiency particulate air filters, can also help minimize exposure to air pollutants that contribute to respiratory cancer risk. Public health programs that raise awareness about these factors and provide practical steps can empower individuals to take proactive measures in daily life and at work.
Diet and physical activity play supportive roles in overall health, and while they do not eliminate the risk of developing cancer, a balanced diet rich in fruits, vegetables, and fiber, alongside regular exercise, can improve immune function, airway health, and resilience during treatment if cancer occurs. Maintaining a healthy weight and controlling chronic conditions such as hypertension and diabetes can also influence a patient’s capacity to tolerate diagnostic procedures and therapies, emphasizing the importance of a comprehensive approach to health maintenance as part of cancer prevention strategies.
Screening programs and access to care
Access to screening programs depends on regional guidelines, insurance coverage, and healthcare infrastructure. People who meet criteria for LDCT screening should discuss eligibility with a clinician who can provide personalized recommendations based on smoking history, age, and overall health. Equitable access is important, as disparities in screening can affect outcomes across different populations. Efforts to expand screening reach often involve public health campaigns, primary care integration, and community based outreach to address barriers such as transportation, language, and mistrust of medical systems.
When you pursue screening, a plan for follow up and communication is essential. Shared decision making ensures that individuals understand the potential benefits and drawbacks, including the chance of false positives and the need for additional testing. Clear documentation of results, a transparent discussion of next steps, and timely referrals to specialists when indicated help maintain patient confidence and reduce anxiety during the screening process. In well organized systems, screening becomes a routine part of preventive care for eligible individuals, enabling earlier detection while respecting patient autonomy.
Access to care also means navigating the emotional and logistical aspects of potential results. Healthcare teams aim to provide compassionate explanations, supportive resources, and practical guidance for scheduling further imaging, biopsies, or consults. Building a trusted relationship with clinicians and care coordinators allows patients to ask questions, express concerns, and participate fully in decisions about evaluation, treatment, and ongoing monitoring as appropriate to their situation.
What to expect during a medical evaluation
A medical evaluation typically begins with a detailed history and physical examination, where clinicians review symptoms, exposure history, family history, and previous imaging results. They may order diagnostic tests such as a chest X ray, LDCT, or other imaging modalities depending on the presentation and risk profile. Blood tests and pulmonary function tests can provide additional context by assessing how the lungs are functioning, which helps clinicians interpret imaging findings in the broader framework of a patient’s health.
During the consultation, clinicians discuss the meaning of any test results, the potential need for procedures such as biopsy, and the possible treatment options if cancer is confirmed. The discussion should also address the expected timeline, potential side effects of procedures, and the impact on daily life. Providers prioritize clear communication, ensuring that patients understand their choices, the rationale for recommended steps, and the expected prognosis associated with different scenarios. This shared understanding helps patients feel supported and engaged in the decision making process.
In many cases, a multidisciplinary team approach is used, bringing together specialists in radiology, medical oncology, thoracic surgery, pathology, and supportive care. Regular team meetings help coordinate care, align on diagnostic accuracy, and plan effective treatment strategies tailored to the individual’s tumor characteristics, comorbidities, and personal preferences. When a cancer diagnosis is established, the team collaborates with the patient to establish a comprehensive plan that considers curative intent, symptom management, and quality of life considerations throughout the course of treatment.
Psychological and social dimensions of screening and diagnosis
Receiving information about potential lung cancer can trigger a range of emotions, including fear, anxiety, relief, or uncertainty. Acknowledging these feelings as a normal part of a serious health concern helps patients seek appropriate emotional support and engage with the medical process more effectively. Counseling services, patient support groups, and education that explains what each step means can reduce distress and empower patients to participate actively in their care decisions. Support from family, friends, and healthcare professionals contributes to resilience during a challenging period.
Social factors such as work obligations, caregiving responsibilities, and financial considerations also influence how individuals navigate screening and treatment. Clinicians recognize these realities and strive to connect patients with resources that address practical needs, including transportation to appointments, financial counseling, and assistance with insurance navigation. A holistic approach to care acknowledges the person behind the illness and seeks to maintain dignity, autonomy, and the ability to pursue an acceptable quality of life while managing health concerns.
Emerging technologies and future directions
In the field of lung cancer detection, research continues to refine imaging techniques, data analysis, and risk stratification. Artificial intelligence and machine learning are being explored to improve the interpretation of scans, reduce false positives, and identify subtle patterns that may indicate early cancer. Researchers are investigating new biomarkers and liquid biopsy approaches that could complement imaging by offering minimally invasive screening options or monitoring tools during and after treatment. These advances hold promise for making early detection more accurate, accessible, and acceptable to a broader segment of the population.
Clinical trials and translational research increasingly focus on integrating screening programs with personalized risk assessments and tailored follow up. By combining patient specific data, imaging characteristics, and molecular information, clinicians aim to create more precise pathways that minimize unnecessary procedures while maximizing the chance of identifying cancers at a curable stage. The evolving landscape emphasizes patient education and empowerment so people can make informed choices about screening, participate in research when appropriate, and benefit from breakthroughs as they become part of standard care.
As knowledge grows, guidelines may adapt to reflect new evidence about the balance of benefits and harms associated with screening and early detection strategies. Ongoing collaboration among researchers, clinicians, policymakers, and patient communities is essential to ensure that advances translate into real world improvements. For individuals, the practical takeaway remains consistent: stay informed about risk factors, discuss screening with trusted clinicians when eligible, and seek evaluation promptly if signs emerge. The combination of informed self care and professional surveillance offers the best chance to detect lung cancer early and improve outcomes.